We share our bodies with trillions of microorganisms. This community of bacteria, fungi, viruses, and parasites is known as the microbiota. “Previous estimates have suggested that there could be ten times as many microbial cells in our body as human cells,” says Naisi Zhao, MHS, DrPH, a research assistant professor at Tufts University School of Medicine Department of Public Health & Community Medicine. The collective genomes of the microbiota are called the microbiome. “Each of us has about 23,000 human genes, and around three million bacterial genes,” says Zhao.
“The gut microbiota starts to establish a stable population early in life,” Zhao explains. “These ‘new microbial friends’ interact with the young immune system in the gut and are established as a typical adult-like gut microbiota by about age three. If a bacterial strain is not a part of your gut microbiota by age three, we’re not yet sure if it will be able to colonize in your gut, no matter how beneficial it is.”
These microorganisms, found mostly in the lower part of the intestines, help digest food, synthesize a variety of vitamins and hormones, and produce metabolites that influence our health.
Microbiome and Health. “We know the gut microbiome plays a fundamental role in multiple aspects of human health,” says Eran Segal, PhD, a professor at the Weizmann Institute of Science who heads a multi-disciplinary team applying computational and systems biology to the microbiome, “including obesity, cardiovascular disease, inflammatory diseases, metabolic diseases, non-alcoholic fatty liver disease, cancer, aging, and neurodegenerative disorders.” Unfortunately, we have yet to figure out exactly how these interactions play out. “The complexity of people’s habitual diets, the difficulty of disentangling the impact of dietary intake from other lifestyle variables, and the personalized nature of the microbiome make this a very difficult topic to study,” says José Ordovás, a professor at the Friedman School and senior scientist and leader of the Nutrition and Genomics Team at the Human Nutrition Research Center on Aging (HNRCA).
We do know our diet and other environmental factors can encourage the growth of some bacteria and discourage others. “We’re shaping this microbial ecosystem as we eat,” says Zhao. “Any undigested food becomes available to our gut bacteria. They feed on nutrients from our leftovers and grow.” What we eat determines which microbes get to flourish, the ‘good,’ or the ‘bad.’ Nourishing the right microbes can benefit our health.
Some of the vitamins produced by our gut microbiota are absorbed into our bloodstreams, just like those we get directly from foods. Other compounds our microbiota make when they break down our undigested food can impact our health in ways we are just beginning to understand. “Some of these byproducts, called metabolites, have physiological effects,” says Meng Wang, PhD, a postdoctoral fellow at the Friedman School conducting research on microbiome metabolites. For example, the short chain fatty acids produced when gut bacteria digest dietary fiber are used as an energy source and may play a role in regulating blood pressure. “Some metabolites, however, can be harmful,” says Wang.
Zhao suggests we think of humans as superorganisms. “It’s not just our own cells that play a role in our health, it’s us plus our microbiome,” she says. “It’s this relationship scientists are hard at work trying to understand.”
Let’s take a look at what we know about how a few common health problems may be impacted by the gut microbiome:
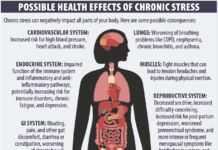
- Systemic Inflammation: Low-grade inflammation throughout the body plays a role in cardiovascular disease and other health problems. Research has suggested a link between gut bacteria and this systemic inflammation. “A certain type of bacteria, called gram-negative, has molecules called lipopolysaccharides (LPS) sticking out of their outer membranes,” says Zhao. “If you have a large population of gram-negative bacteria, these LPS molecules can induce a strong immune reaction that triggers low-grade inflammation.” In one early study, LPS molecules injected into healthy animals caused them to develop low-grade inflammation and insulin resistance. While this animal study does not prove the same thing happens in humans, it suggests gut bacteria might play a role in systemic inflammation as well as weight and blood sugar control. “Molecules our gut bacteria generate could enter our bloodstream, where they likely cause some kind of reaction,” says Zhao.
- Obesity: “Microbiome links to obesity have attracted a lot of interest,” says Ordovás, “although results in human studies have varied.” In 2006, scientists transplanted microbiota from genetically obese mice or normal lean mice into germ-free mice (raised to harbor no microbes at all). Although they were all fed the same chow diet, those with the microbiota from obese mice became fatter compared to those who had received transplants from normal weight mice.
In a case study in China, a morbidly obese individual was fed a high fiber diet to encourage the growth of “good” bacteria. “The individual lost a significant amount of weight and had a lot of really positive health changes,” says Zhao, who was close to the study. “Genetic sequencing of his stool found a specific bacteria strain in very high abundance before the diet had been reduced to almost undetectable levels after the intervention period.” While this is just one individual, this particular study helped to address the “chicken/egg” problem that accompanies a lot of nutrition research: does changing the microbiota lead to improved health outcomes, or does better health change the microbiome? “In this case study, the bacterial strain from the obese individual’s microbiota was isolated and inoculated into germ-free mice,” Zhao explains. The mice experienced changes related to fat burning and fat storage and they developed systemic inflammation and insulin resistance. “This finding points to causation: the pre-diet microbiome was negatively impacting health and weight,” says Zhao.
These fascinating studies do not prove the microbiome causes obesity in humans, nor that fecal transplants can decrease body weight. In fact, the few human studies available have been negative.
Probiotics are live bacteria or other microorganisms added to foods and beverages or packaged in pills with the goal of improving the composition of your gut microbiota. They have become popular with food manufacturers and consumers—popping up in everything from yogurts to sweets.
Unfortunately, we do not yet know which organisms are the most beneficial, how to effectively deliver them through the stomach to the intestines, and whether or not they will be able to colonize the gut.
Eating a plant-based diet rich in fruits, vegetables, nuts/seeds, beans, and whole grains is much more likely to benefit your gut bacteria than supplementing with probiotics.
- Muscle: There is a growing body of research in animal models suggesting the gut microbiome influences muscle, including mass, strength, and aerobic endurance capacity, but there’s not much data in humans. “A small number of studies have looked at the differences in gut microbiome composition between highly fit and lesser fit individuals,” says Michael Lustgarten, PhD, a scientist on the Nutrition, Exercise Physiology, and Sarcopenia Team at the HNRCA, “and two randomized controlled trials have demonstrated that exercise training improved muscle function in conjunction with changes in microbiome composition in older adults, but we don’t yet know if the microbiome on its own can impact muscle-related measures in people.”
Any affect that the microbiome has on muscle would likely be due to metabolites such as the short chain fatty acids that are produced when bacteria digest soluble fiber. “Most people in the U.S. don’t consume the recommended 20 to 30 grams of fiber a day,” says Lustgarten, “and they certainly don’t get enough soluble fiber from foods like fruits, vegetables, legumes, and whole grains.” Older adults are even less likely to meet their fiber needs, and they are particularly at risk for loss of muscle mass and infiltration of fat into muscle.
- Cardiovascular Disease: The output of some gut microbes may play a role in increasing risk for cardiovascular disease (CVD). The most studied example of this possible connection is a molecule called TMAO (trimethylamine N-oxide). When we ingest phosphatidyl choline (found in a variety of animal products) and carnitine (almost exclusively from red meat), gut microbes turn these nutrients into a molecule called TMA, which is then absorbed and converted in the liver to TMAO. High levels of TMAO in the blood are associated with increased risk for cardiovascular disease. Studies have demonstrated TMAO can promote atherosclerosis, thrombosis (blood clot formation), and hyperglycemia (high blood sugar) in animal models. “There seems to be an association,” says Wang, “but evidence from human studies has not been sufficient to establish a causal relationship between TMAO and cardiovascular disease.”
“The possibility that TMAO influences cardiovascular health would help explain the association between red meat intake and CVD,” Wang suggests. “We know red meat intake is associated with increased CVD risk, but we don’t fully understand the mechanism behind the connection. While it’s just a hypothesis at this time, it could be that gut microbes metabolizing the carnitine in red meat leads to increased TMAO, which increases CVD risk.”
Two randomized controlled trials recently showed that TMAO levels in the body rose in people at high cardiometabolic risk after they consumed foods rich in omega-3 fatty acids (found in fish) and whole-grain cereals, both of which are associated with lower CVD risk. This finding casts doubt on the usefulness of TMAO as a universally valid biomarker of cardiometabolic risk. More research is needed.
Other possible links between diet, the microbiome, and cardiometabolic health are being explored. Ordovás and his colleagues recently published a study that shows a strong link between the gut microbiome and markers of cardiometabolic health like blood pressure, triglyceride levels, total cholesterol, HDL (good) cholesterol, LDL (bad) cholesterol, and blood sugar levels. This study, called Personalised Responses to Dietary Composition Trial (PREDICT 1), included more than 1,000 participants in the U.K. and the U.S. who spent a day in the lab eating specific meals and being tested to determine their individual responses. Participants also carefully tracked dietary intake, took blood tests, gave stool samples for gut microbiome analysis, and underwent continuous glucose monitoring for two weeks. The researchers mapped out what affect foods had on the composition of the microbiome. They found a panel of 15 bacterial strains all consistently associated with markers of good health and 15 consistently associated with markers of poor health. “These same microbes were also associated with certain dietary intake,” says Ordovás. In other words, a healthy dietary pattern was associated with higher levels of specific species of bacteria, and people with more of these species were likely to have lower blood pressure, better blood sugar control, and other important measures of health. Less healthy dietary intake was associated with higher levels of other bacteria species, and the presence of these species was associated with markers of poor cardiometabolic health.
Scientists can take a sample of your stool and do genetic sequencing to determine the makeup of your gut microbiome, but—contrary to what you may hear from testing company ads—there’s not a lot anyone can do with that information at this time.
The general consensus is that we just don’t know enough about how a food or food component will impact a particular strain of bacteria, or how that reaction might differ when that bacterial strain is in the company of the whole gut microbiota. Other factors, including your activity level, the environment, sleep, stress, and your genes, impact your microbiome—and your health. We don’t even know the function of 40 percent of the estimated 10 million bacterial genes that can be found in our microbiome. All of this adds up to a high level of uncertainty when it comes to trying to improve your health by manipulating your gut microbiome.
There are some companies that couple microbiome testing with other measures such as continuous glucose monitoring and blood testing to try to get a more nuanced picture of your personal needs. While there are some promising results, this field is still in its infancy. You may want to save your money until we learn more. Meanwhile, including plenty of unprocessed and minimally processed plant-based foods and fermented choices in your diet is good for your health—and the health of your microbial companions.
Having a greater variety of bacteria in the gut was also found to be beneficial. “The richer the diversity of species in the gut, the less likely participants were to have high BMI, fatty liver, or high levels of visceral fat,” says Ordovás, “and the more likely they were to have higher HDL (good) cholesterol levels.” This study is the first to identify microbial signatures that are related to both diet and metabolic health. “These results will help us begin to use the gut microbiome as a biomarker for cardiometabolic risk,” says Ordovás.

Image © marilyna | Getty Images
Shaping Your Microbiome. “Diet is probably the most powerful force shaping our gut microbiota,” says Zhao. “We are feeding ourselves and the bacteria in our gut with every meal we eat.” If we can understand the health impacts of different gut microbes and learn what they like to eat, we should be able to improve our health by manipulating our microbiome. “The gut microbiota is an ecosystem,” says Zhao. “If we can make sure the good bacteria are provided with nutrients to grow and flourish, they will help keep the bad ones in check.” Research has shown that, in general, plant-based diets are associated with greater microbial diversity than animal-based diets, and higher diversity is associated with better health outcomes. Not all plant-based foods are equally health-promoting, however.
We still have much to learn about our personal microbiomes and how to manipulate them to maximize health. For now, try these tips:
- Nourish your inner world. Diets high in healthy plant-based and fermented foods encourage the proliferation of health-promoting gut microbes. Enjoy unprocessed and minimally processed fruits, vegetables, nuts/seeds, legumes, and whole grains, as well as yogurt, kefir, kambucha, and kimchi.
- Skip the probiotic supplements. We do not yet know which organisms are the most beneficial, how to effectively deliver them through the stomach to the intestines, and whether or not they will be able to colonize the gut.
- Hold off on testing. A few reputable companies are on the right track, but personalized nutrition to adjust our microbiome for health is in its infancy.
Dietary fiber is very important. The best way to get fiber is to eat plants—fruits, vegetables, nuts/seeds, beans/lentils, and whole grains. “Most ‘good’ bacteria living in our gut feed on fiber,” says Zhao. “We cannot digest fiber, so it provides the microbes with plenty to eat.”
In the PREDICT 1 study, healthier plant-based foods (like spinach, seeds, tomatoes, and broccoli) were more strongly associated with positive microbial strains than less healthy plant-based foods (for example, juices, sweetened beverages, and refined grains). The same was true for animal-based foods: foods generally considered to be healthy (for example fish and eggs) correlated with “good” bacterial strains, while less healthy choices (like meat pies, bacon, and dairy desserts) correlated more strongly with the presence of “bad” strains. In another recent study, intake of plant- and fiber-rich dietary choices was associated with a greater variety of gut microbes, which, as explained above, is considered beneficial.
Fermented foods also nourish gut microbiota. A recently published study found that a 10-week diet rich in fermented foods like yogurt, kefir (a yogurt drink), miso, tempeh, kambucha tea, and kimchi (Korean fermented cabbage) and other fermented vegetables enhanced the diversity of gut microbes. Participants on the high fermented foods diet also had lower levels of inflammatory proteins in their blood. (Do watch out for high levels of sodium or added sugars in some fermented foods.)
“It’s challenging to disentangle the independent associations of single foods with microbial species,” says Ordovás, “but a diversity of healthy foods, including plenty of healthy plant-based foods, is proving to be important.”

Image © thesomegirl | Getty Images
Looking to the Future. While the research discussed above is promising, it is still speculative at this time. “Looking at the microbiome should capture different aspects of disease than the blood tests and screenings we currently use,” says Segal. “A combination of methods could lead to earlier and more robust disease detection. However, it must be stressed that very few microbiome-based markers predictive of disease onset and progression have been found to date, and none are currently used by healthcare systems. Discovery of microbiome-based risk factors is a promising research area, but it is still in its infancy.” Zhao agrees. “We hope the microbiome can someday be used to personalize nutrition,” she says. “We’d ideally be able to take a stool sample before and after a diet as a way to measure how that diet has impacted the individual’s health. We’re not quite there yet, but that’s the goal we’re working towards.”